

ZUBSOLV: Clinically comparable to Suboxone® film with the addition of patient-preferred attributes1,2
The largest combination buprenorphine/naloxone opioid dependence trial for efficacy and safety conducted in the United States to date.
Actor portrayal
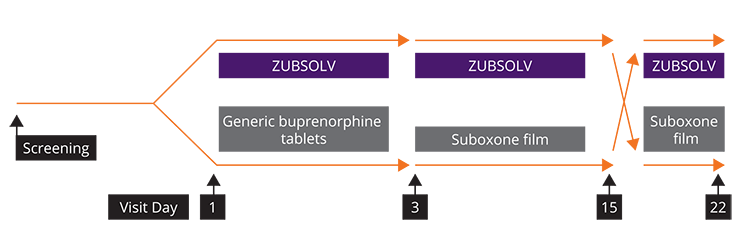
Induction, Stabilization, Adherence and Retention Trial1
- A randomized, noninferiority study to assess early treatment efficacy of ZUBSOLV versus Suboxone film and to explore switching between treatments
- Opioid dependent adult male or female subjects ≥18 to 65 years of age
- 758 subjects randomized
- Stabilization doses were titrated to a maximal daily dose of 17.1 mg/4.2 mg and 24 mg/6 mg for ZUBSOLV and Suboxone, respectively, based upon clinical symptoms
PRIMARY OBJECTIVES:
Retention in treatment after induction at Day 3 and stabilization at Day 15
Secondary OBJECTIVES:
Assessment of treatment effects on:
- Opioid withdrawal symptoms (COWS, SOWS)
- Opioid cravings (Craving VAS)
- Illicit drug use (UDS, SRSU)
- Addiction severity (CGI-S, CGI-I, PGI-I)
Formulation acceptability and preference of ZUBSOLV compared to Suboxone film
Switching between Suboxone film and ZUBSOLV
Safety and tolerability of ZUBSOLV
COWS (Clinical Opiate Withdrawal Scale)
SOWS (Subjective Opiate Withdrawal Scale)
VAS (Visual Analogue Scale)
UDS (Urine Drug Screen)
SOWS (Subjective Opiate Withdrawal Scale)
VAS (Visual Analogue Scale)
UDS (Urine Drug Screen)
SRSU (Self Report of Substance Use)
CGI-S (Clinical Global Impression-Severity)
CGI-I (Clinical Global Impression-Improvement)
PGI-I (Patient’s Global Impression-Improvement)
CGI-S (Clinical Global Impression-Severity)
CGI-I (Clinical Global Impression-Improvement)
PGI-I (Patient’s Global Impression-Improvement)
Actor portrayal
Patient Demographics
Study Participants
- 758 adult patients
- Aged 18 to 65 years (mean 35.6 years)
- Meeting DSM-IV-TR® criteria for opioid dependence
- Mean duration of opioid dependence: 10.6 years
Male
Female
ZUBSOLV showed no increased rate of withdrawal symptoms or opioid cravings compared to Suboxone film1
More than 70% of patients preferred ZUBSOLV’S attributes to Suboxone film2
Significantly more patients preferred ZUBSOLV to Suboxone film based on overall preference, taste, mouthfeel, and ease of administration
Adverse events were similar across treatments1,3
Treatment-related adverse events reported in >2% of patients for Days 1-15. Data are given as number (%) of patients.
MedDRA Preferred Term
Double-blind period (Days 1-2)
Any
Headache
Vomiting
Nausea
Dry mouth
Open-label period (Days 3-15)
Any
Constipation
Headache
ZUBSOLV Sublingual Tablets
n = 383
61 (15.9)
20 (5.2)
12 (3.1)
8 (2.1)
8 (2.1)
n = 357
42 (11.8)
10 (2.8)
5 (1.4)
Generic Buprenorphine Tablets/ Suboxone Film
n = 375
55 (14.7)
19 (5.1)
11 (2.9)
15 (4.0)
2 (0.5)
n = 344
37 (10.8)
12 (3.5)
7 (2.0)
All Patients
N = 758
116 (15.3)
39 (5.1)
23 (3.0)
23 (3.0)
10 (1.3)
N = 701
79 (11.3)
22 (3.1)
12 (1.7)
- Gastrointestinal effects were minimal with constipation reported in <5% of patients in the study. Similar improvements were demonstrated in constipation symptoms from baseline to Day 15 in both groups
- Nervous system disorders were similar between groups, all were mild to moderate in severity with headache being the most commonly reported event
- 99% of treatment-related TEAEs were of mild or moderate severity
- No deaths occurred during the study
Conclusions
ZUBSOLV patients demonstrated comparable clinical efficacy vs Suboxone film
When changed from Suboxone film, no increased rate of withdrawal symptoms, opioid cravings, or treatment discontinuation was observed
After experiencing both ZUBSOLV and Suboxone film, more than 70% of patients preferred the attributes of ZUBSOLV overall (P<0.0001)
Treatment-related adverse events were similar for ZUBSOLV and generic buprenorphine tablets/Suboxone film
Actor portrayal
In an open-label, comparative bioavailability study in 60 healthy volunteers*:
ZUBSOLV offers a highly efficient delivery system to support your patients on their journey to recovery4
ZUBSOLV delivered comparable blood concentration to other buprenorphine/naloxone tablets, but with less drug4
- ZUBSOLV has an advanced formulation, which contains less buprenorphine than other buprenorphine/naloxone-containing sublingual tablets to achieve equivalent systemic exposure†
dosing flexibility
*Study design: Open-label, fasting, randomized, 2-period, crossover, comparative bioavailability study in 60 healthy volunteers. Subjects received either ZUBSOLV sublingual tablet or Suboxone sublingual tablet under naltrexone block according to a 2-sequence randomization schedule. Blood samples were collected at predose and at intervals over 72 hours after dosing each period. Pharmacokinetics, tolerability, safety, and dissolve time were assessed and acceptability and subject preference of drug product were evaluated by specific questioning.
†Exposure is the concentration of buprenorphine detectable in the plasma evaluated after dosing.
†Exposure is the concentration of buprenorphine detectable in the plasma evaluated after dosing.
References:
- Gunderson EW, Hjelmström P, Sumner M. Effects of a higher-bioavailability buprenorphine/naloxone sublingual tablet versus buprenorphine/naloxone film for the treatment of opioid dependence during induction and stabilization: A multicenter, randomized trial. Clin Ther. 2015;37(10):2244-55.
- Gunderson EW, Sumner M. Efficacy of buprenorphine/naloxone rapidly dissolving sublingual tablets (BNX-RDT) after switching from BNX sublingual film. J Addict Med. 2016;10(2):122-8.
- Data on file. Dexcel Pharma USA.
- Fischer A, Jonsson M, Hjelmstrom P. Pharmaceutical and pharmacokinetic characterization of a novel sublingual buprenorphine/naloxone tablet formulation in healthy volunteers. Drug Dev Ind Pharm. 2015;41(1):79-84.
Indication
ZUBSOLV® (buprenorphine and naloxone) sublingual tablet (CIII) is indicated for the treatment of opioid dependence. ZUBSOLV should be used as part of a complete treatment plan that includes counseling and psychosocial support.
Important Safety Information
Contraindications
- ZUBSOLV is contraindicated in patients with a history of hypersensitivity to buprenorphine or naloxone, as serious adverse reactions, including anaphylactic shock, have been reported.
Warnings and Precautions
- Addiction, Abuse, and Misuse: Buprenorphine can be abused in a similar manner to other opioids. Monitor patients for conditions indicative of diversion or progression of opioid dependence and addictive behaviors. Multiple refills should not be prescribed early in treatment or without appropriate patient follow-up visits.
- Risk of Life-Threatening Respiratory and Central Nervous System (CNS) Depression: Life-threatening respiratory depression and death have occurred in association with buprenorphine use. Warn patients of the potential danger of self-administration of benzodiazepines or other CNS depressants while under treatment with ZUBSOLV.
- Patient Access to an Opioid Overdose Reversal Agent for the Emergency Treatment of Opioid Overdose: Inform patients and caregivers about opioid overdose reversal agents (e.g., naloxone, nalmefene). Strongly consider recommending or prescribing an overdose reversal agent for the emergency treatment of opioid overdose, both when initiating and renewing treatment with ZUBSOLV, and consider recommending or prescribing such an agent if the patient has household members (including children) or other close contacts at risk for accidental ingestion or opioid overdose.
- Advise patients and caregivers that opioid overdose reversal agents, such as naloxone or nalmefene, may also be administered for a known or suspected overdose with ZUBSOLV itself.
- Educate patients and caregivers on how to recognize respiratory depression, and how to use an opioid overdose reversal agent for emergency treatment. Emphasize the importance of calling 911 or getting emergency help, even if an opioid overdose reversal agent is administered.
- Managing Risks from Concomitant Use of Benzodiazepines or Other CNS Depressants: Concomitant use of buprenorphine and benzodiazepines and/or other CNS depressants (e.g., alcohol, non-benzodiazepine sedative/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids [gabapentin or pregabalin], and other opioids) increases the risk of adverse reactions including overdose, respiratory depression and death. As a routine part of orientation to buprenorphine treatment, educate patients about the risks of concomitant use of benzodiazepines, sedatives, opioid analgesics, and alcohol. Develop strategies to manage use of prescribed or illicit benzodiazepines or other CNS depressants at initiation of buprenorphine treatment, or if it emerges as a concern during treatment.
- Before co-prescribing benzodiazepines, ensure that patients are properly diagnosed and consider alternative treatments to address anxiety or insomnia.
- Take measures to confirm that patients are taking their medication as prescribed and are not diverting or supplementing with illicit drugs, including toxicology screening to test for prescribed and illicit benzodiazepines.
- Unintentional Pediatric Exposure: Store ZUBSOLV safely out of the sight and reach of children. Buprenorphine can cause severe, possibly fatal respiratory depression in children.
- Neonatal Opioid Withdrawal Syndrome (NOWS): Neonatal opioid withdrawal syndrome (NOWS) is an expected and treatable outcome of prolonged use of opioids during pregnancy.
- Adrenal Insufficiency: If diagnosed, treat with physiologic replacement of corticosteroids, and wean patients off of the opioid.
- Risk of Opioid Withdrawal with Abrupt Discontinuation: If treatment is temporarily interrupted or discontinued, monitor patients for withdrawal and treat appropriately.
- Risk of Hepatitis; Hepatic Events: Monitor liver function tests prior to initiation and during treatment and evaluate suspected hepatic events.
- Precipitation of Opioid Withdrawal Signs and Symptoms: An opioid withdrawal syndrome is likely to occur with parenteral misuse of ZUBSOLV by individuals physically dependent on full opioid agonists or by sublingual administration before the agonist effects of other opioids have subsided.
- Risk of Overdose in Opioid-Naïve Patients: ZUBSOLV is not appropriate as an analgesic. There have been reported deaths of opioid-naïve individuals who received a 2-mg sublingual dose of buprenorphine.
- Dental Adverse Events: Cases of dental caries, some severe (i.e., tooth fracture, tooth loss), have been reported following the use of transmucosal buprenorphine-containing products. Educate patients to seek dental care and strategies to maintain or improve oral health while being treated with ZUBSOLV.
- QTc Prolongation: Thorough QT studies with buprenorphine products have demonstrated QT prolongation ≤15 msec. The risk of combining buprenorphine with other QT prolonging agents is not known. Consider these observations in clinical decisions when prescribing ZUBSOLV to patients with QT-related risk factors.
Use in Specific Populations
- Lactation: Buprenorphine passes into mother’s milk.
- Geriatric Patients: Monitor for sedation and respiratory depression.
- Moderate and Severe Hepatic Impairment: Buprenorphine/naloxone products are not recommended in patients with severe hepatic impairment and may not be appropriate for patients with moderate hepatic impairment.
Adverse Reactions & Drug Interactions
- Adverse events commonly observed with the sublingual administration of ZUBSOLV are headache, nausea, vomiting, hyperhidrosis, constipation, signs and symptoms of withdrawal, insomnia, pain, and peripheral edema.
- Benzodiazepines: Use caution in prescribing ZUBSOLV for patients receiving benzodiazepines or other CNS depressants and warn patients against concomitant self-administration/misuse.
- CYP3A4 Inhibitors and Inducers: Monitor patients starting or ending CYP3A4 inhibitors or inducers for potential over or under dosing.
- Antiretrovirals: Patients who are on chronic buprenorphine treatment should have their dose monitored if NNRTIs are added to their treatment regimen. Monitor patients taking buprenorphine and atazanavir with and without ritonavir, and reduce dose of buprenorphine if warranted.
- Serotonergic Drugs: Concomitant use may result in serotonin syndrome. Discontinue ZUBSOLV if serotonin syndrome is suspected.
This is not a complete list of potential adverse events associated with buprenorphine/naloxone tablets. For additional safety information, please see Full Prescribing Information
Indication
See More
ZUBSOLV® (buprenorphine and naloxone) sublingual tablet (CIII) is indicated for the treatment of opioid dependence. ZUBSOLV should be used as part of a complete treatment plan that includes counseling and psychosocial support.
Important Safety Information
Contraindications
- ZUBSOLV is contraindicated in patients with a history of hypersensitivity to buprenorphine or naloxone, as serious adverse reactions, including anaphylactic shock, have been reported.
This site is intended for residents of the United States.
ZUBSOLV is a licensed trademark of Dexcel Pharma USA.
Other trademarks, registered or otherwise, are the property of their respective owner(s).
This site is sponsored by Dexcel Pharma USA.
© 2026 Dexcel Pharma USA. All rights reserved.